510(k) Submission Specialist for Medical Devices
-
Delivery Time2 Days
-
English levelConversational
-
LocationUnited States
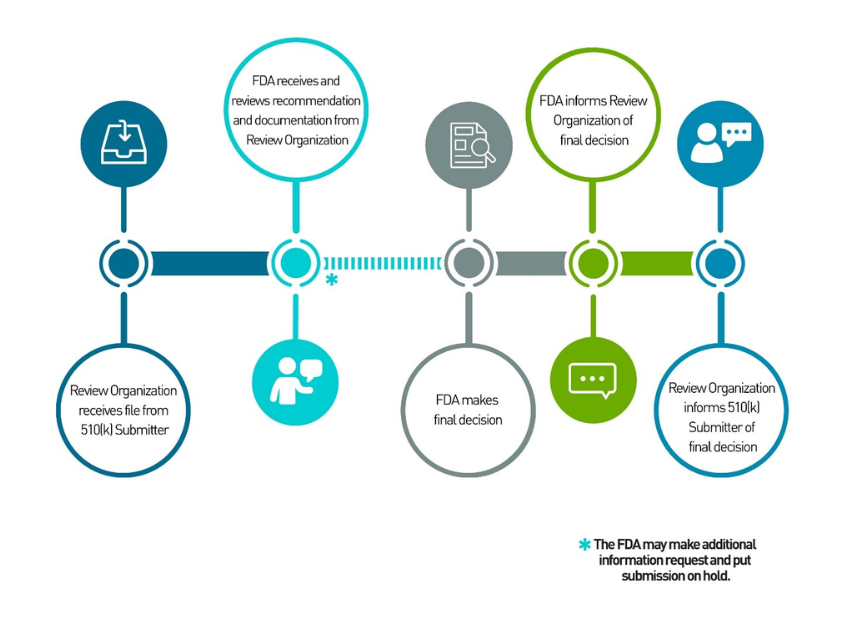
Service Description
What methods of payments are supported?
We support various methods of payment for your convenience, including major credit cards, bank transfers, and online payment platforms such as PayPal. If you have a preferred method of payment that is not listed, please feel free to discuss it with us.
Can I cancel at anytime?
Yes, you can cancel the service at any time. However, please note that any work already completed at the time of cancellation will be billed accordingly. If you cancel before work has commenced, you will receive a full refund of any deposit paid.
How do I get a receipt for my purchase?
Once a payment is made, an official receipt will be issued and sent to you via email. If you require a hard copy, please let us know, and we can arrange for one to be mailed to you.
How do I pay the payments?
Payments are structured to support the progress of your 510(k) submission process. An initial 50% deposit is required to commence work. Once the documents are filed with the FDA, an additional 25% payment is due. The final 25% is payable just before we receive the FDA approval. This staged payment process is designed to align with key milestones in your submission journey and to provide you with peace of mind as we move forward.








Agent Pakulla in-depth knowledge and meticulous attention to detail were pivotal in our successful 510(k) submission. Their guidance through the complex regulatory landscape ensured a smooth and efficient process. Highly recommend for anyone looking to bring medical devices to market!